|
– Access at a full resolution of this Table as PDF (made available by King’s Center for Visualization in Science).īy virtue of its work in relation with the chemical elements, IUPAC can dispense a periodic table that is up-to-date. – Review the latest IPTEI element-by-element review including a chart of all known stable and radioactive isotopes for each element and examples practical applications of isotopic measurements and technologies – Explore the interactive version at /isotopes-matter (or see release) – Read “Atomic Weights: No Longer Constants of Nature”, Chem Int 33(2), 10–15 (2011), IUPAC Periodic Table of the Elements and Isotopes (IPTEI) for the Educational Community Coplen, Peter Mahaffy, Ian Mills, Roberto Marquardt, and more. Leigh, Sigurd Hofmann, Eric Scerri, Juris Meija, Norman E. See PAC (AOP ) for full details or visit Commission II.1 ĭownload the PDF version (letter size or A4) or A3 (PDF) or see earlier versionsĬheck out SPECIAL Chem Int Jan 2019 - International Year of the Periodic Table (IYPT) - with contributions by Jan Reedijk, Natalia Tarasova, G.J. For elements that lack isotopes with a characteristic isotopic abundance in natural terrestrial samples, the mass number of the nuclide with the longest confirmed half-life is listed between square brackets. These noble gases are highly stable and do not react to form compounds under normal conditions.ģ) A column of elements in the periodic table.The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights ( CIAAW), compiled as part of the 2021 Table of Standard Atomic Weights 2021. Elements Group 18 have a complete outer shell with eight electrons. They are also very reactive because they have seven electrons in the outer shell and will readily accept an electron in order to reach the ion configuration with the ideal number of eight electrons in the outer shell. They all have seven electrons in the outer orbital (two in level s and five in level p). :max_bytes(150000):strip_icc()/periodic-table-of-elements-680789917-58ea3e903df78c5162f92b6f.jpg)
They have only one electron in the outer shell (valence electron) and as a result are highly reactive. All elements in Group 1 for instance are alkali metals. Elements are arranged in these groups according to whose proprieties are similar. Because of the confusion the old IUPAC and the CAS system created, the IUPAC adopted their new system. This method, however, labeled columns 1 and 2 as 'IA' and 'IIA', columns 3 through 7 as 'IIIB' through 'VIB', column 8 through 10 as 'VIII', columns 11 and 12 as 'IB' and 'IIB' and columns 13 through 18 as 'IIIA' through 'VIIIA'. The CAS system, which also used Roman numerals followed by an 'A' or 'B'. Columns were numbed such that columns one through seven were numbered 'IA' through 'VIIA', columns 8 through 10 were labeled 'VIIIA', columns 11 through 17 were numbered 'IB' through 'VIIB' and column 18 was numbered 'VIII'. The old IUPAC system, which labeled columns with Roman numerals followed by either the letter 'A' or 'B'. To reduce confusion caused by the other two systems, this is the system that is used in articles on this web site.  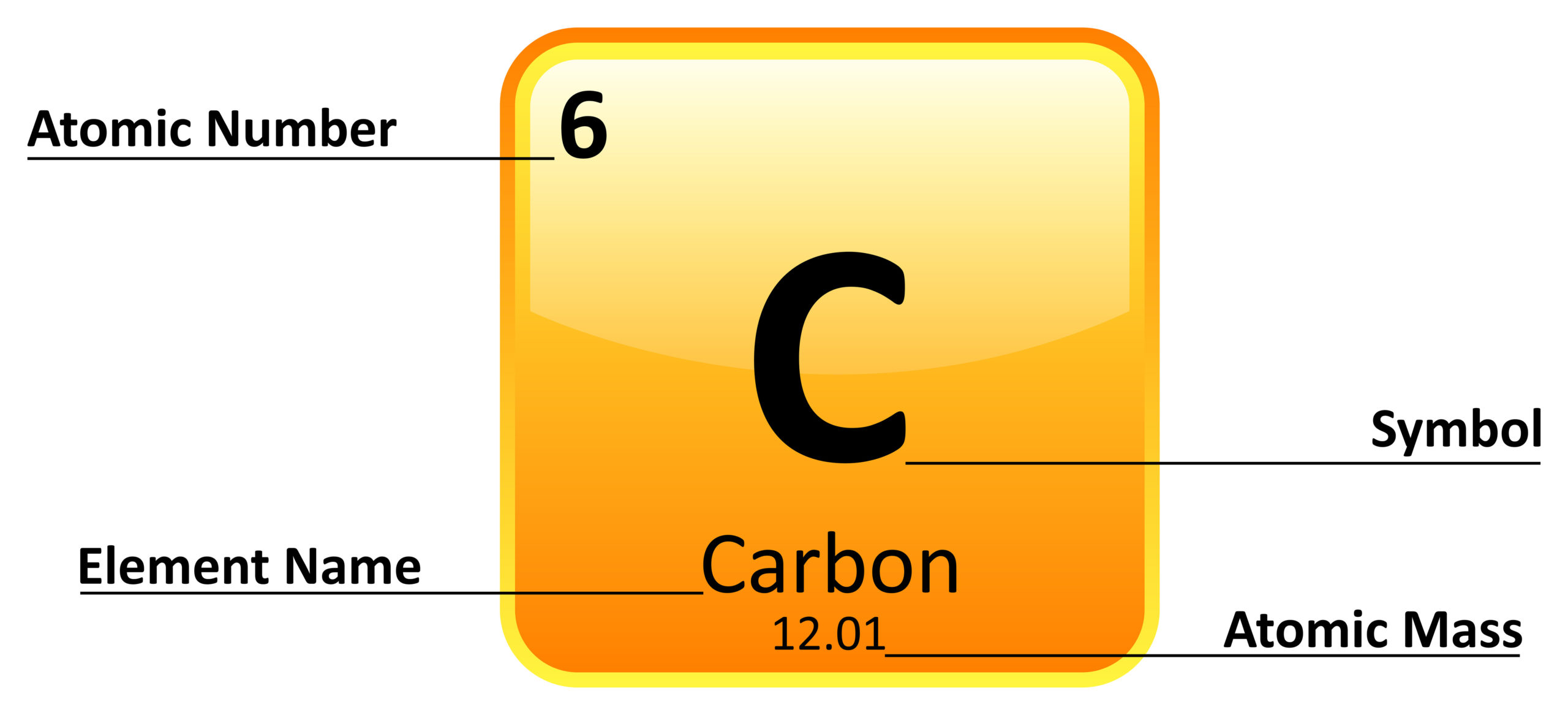
There are three common numbering systems for these groups: The new IUPAC system, which numbers each column with Arabic numbers from 1 (one) through 18 (eighteen). For more information see Periodic TablesĢ) The vertical columns (major classes or divisions) into which elements are arranged in the periodic table of elements. 1) A vertical column in the periodic table also called a family.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
